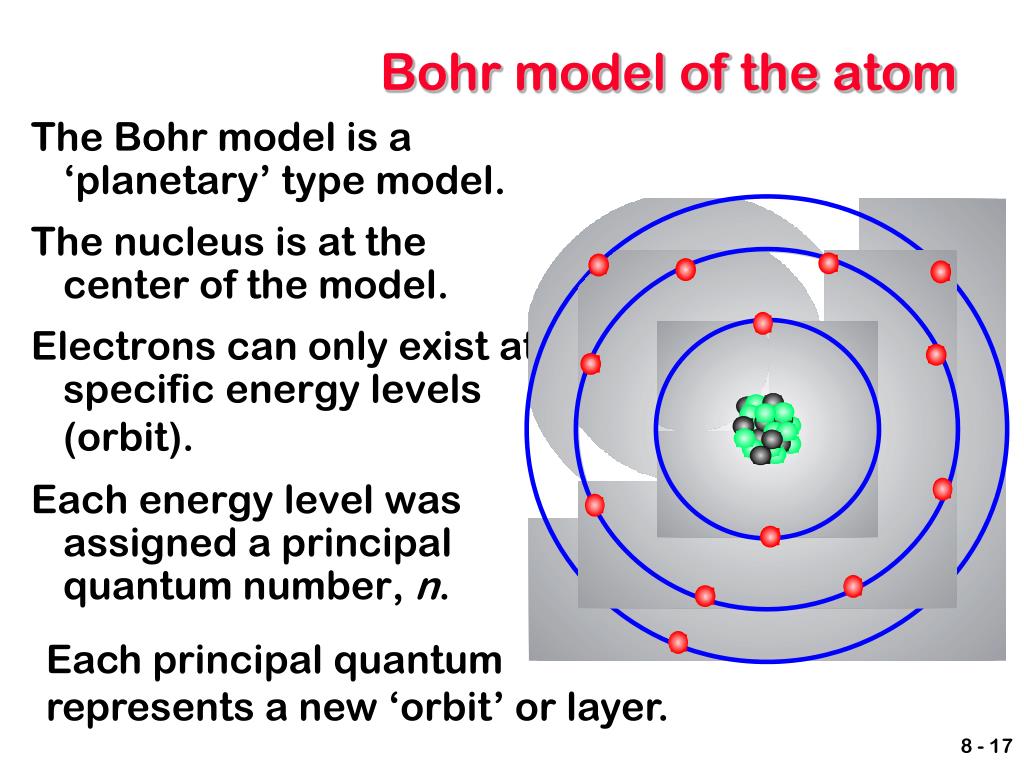
Learn the postulates, limitations and examples of Bohr's model with videos, FAQs and numerical problems. Also, despite a great deal of tinkering, such as assuming that. The popular theory of atomic structure at the time of Rutherford’s experiment was the plum pudding model. Learn how Bohr models are used to represent atoms and their energy levels. 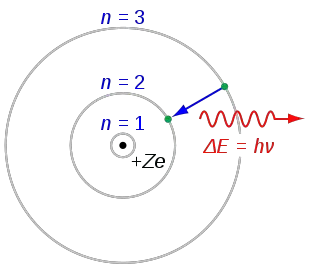
The following are his key contributions to our understanding of atomic structure: Unfortunately, Bohr could not explain why the electron should be restricted to particular orbits. The gold foil experiment was conducted under the supervision of Ernest Rutherford at the University of Manchester in 1909 by Hans Geiger and Ernest Marsden. Bohrs model calculated the following energies for an electron in the shell, n : E (n) 1 n 2 13. Bohrs model of hydrogen is based on the nonclassical assumption that electrons travel in specific shells, or orbits, around the nucleus. It was proposed by Neil Bohr in 1915 and modified by Rutherford's model. Bohr’s model of the hydrogen atom gave an exact explanation for its observed emission spectrum. How Bohrs model of hydrogen explains atomic emission spectra. Check how the prediction of the model matches the experimental results. Bohr's model of an atom is a theory that explains that electrons move in fixed orbits around the nucleus and have a fixed energy. L is defined to be r x p, which is rpsin (theta), where theta is the angle between the radius vector and the momentum vector. How did scientists figure out the structure of atoms without looking at them Try out different models by shooting light at the atom. Where \( \Re \) is the Rydberg constant, h is Planck’s constant, c is the speed of light, and n is a positive integer corresponding to the number assigned to the orbit, with n = 1 corresponding to the orbit closest to the nucleus. Check how the prediction of the model matches the experimental results.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
